Abstract:
Objective: Research on the preparation of puerarin capsule and the establishment of its in vitro Analytical Method. Method: Ultraviolet spectrophotometry method is adopted to analyze the impact of different excipients on dissolution rate of puerarin capsule. Results: The dissolution rate of puerarin capsule is optimal when the excipient formula consists of micro crystal cellulose (MCC), PEG-4000 and micronization silica gel. The in vitro accumulative dissolution rate reaches NLT 85% within 45 minutes. The UV standard curve shows a good linearity range between 1.0 - 10.0 µg/mL and the recovery rate is between 98.60% - 101.91%, RSD = 1.95%. Conclusion: The puerarin phosphatide capsule in this research has a good in vitro dissolution, which can be used as an in vivo assessment of the bioavailability to some extent. The ultraviolet spectrophotometry analytical method is easy to operate, fast, precise and accurate, which can be used as an in vitro analytical method for puerarin capsule.
1. 引言
葛根素(Puerarin)是常用中药葛根(Pueraria Lobata Ohwi)的主要有效成分,是一种黄酮类化合物,具有扩张冠状动脉和脑血管、降低心肌氧耗、改善心肌收缩功能、降低血液粘度、增加脑血流量等药理作用 [1] [2] [3] 。由于葛根素结构中的多苯环和多羟基的原因,其水溶性较小(属微溶于水的药物)并且脂溶性也小(难溶于氯仿),难以被胃肠道等生物膜吸收,口服生物利用度低,从而限制了葛根素在临床上的应用,目前临床上只有葛根素注射剂一个剂型。随着葛根素注射剂在临床上的广泛应用,其不良反应的报道逐渐增多,常见的不良反应有变态反应、溶血性贫血、药物热、药疹,而溶血性贫血和过敏性休克严重时可导致死亡 [4] [5] 。为克服这一临床用药难题,药学研究者通过葛根素与磷脂形成复合物,改变其理化性质,从而提高了口服生物利用度 [6] [7] 。本文经过进一步研究,以体外溶出速率为指标对处方进行筛选,制备葛根素胶囊,以满足临床用药的需求。
2. 仪器与材料
2.1. 仪器
电子天平(梅特勒-托利多),恒温磁力搅拌器(上海梅颖浦仪器仪表制造有限公司),旋转蒸发仪(埃朗科技国际贸易(上海)有限公司),真空干燥箱(上海实验仪器厂有限公司),溶出试验仪(天津市天大天发科技有限公司),可见-紫外分光光度计(日本岛津公司)。
2.2. 材料
葛根素对照品(中国食品药品检定研究院,批号110752-201313),葛根素原料(青岛金峰制药有限公司,批号20140612),大豆卵磷脂(美国嘉吉公司,批号129065),交联聚乙烯吡络烷酮(安徽山河药用辅料股份有限公司,批号20140211),羧甲基淀粉钠(安徽山河药用辅料股份有限公司,批号20141228),微晶纤维素(日本旭化成公司,批号6422),聚乙二醇4000(美国陶氏化学公司,批号2K1755S7B1),糊精(安徽山河药用辅料股份有限公司,批号20150123),微粉硅胶(安徽山河药用辅料股份有限公司,批号20141112)。
无水乙醇(天津市富宇精细化工有限公司),磷酸二氢钾(广东光华科技股份有限公司),氢氧化钠(西陇化工股份有限公司),纯化水。
3. 方法与结果
3.1. 葛根素磷脂复合物的制备
称取葛根素原料10 g,大豆卵磷脂12 g,加入100 mL无水乙醇,30℃水浴搅拌反应0.5 h,减压蒸除反应溶剂,50℃真空干燥箱干燥,过40目筛整粒,加适量的辅料,填充胶囊,即得,每粒胶囊含葛根素100 mg。
3.2. 体外分析方法的建立
3.2.1. 对照品溶液的制备
精密称取葛根素对照品10.50 mg (含量以95.5%计)至20 mL容量瓶,加磷酸盐缓冲液(pH6.8)溶解并稀释至刻度,作为对照品溶液备用。
3.2.2. 测定波长的选择
吸取葛根素对照品溶液0.6 mL置50 mL量瓶中,用磷酸盐缓冲液(pH6.8)稀释至刻度,摇匀,以磷酸盐缓冲液(pH6.8)作空白对照,在200~400 nm波长内扫描,结果在251 nm处葛根素有最大吸收,扫描图谱见图1。
3.2.3. 标准曲线
精密吸取葛根素对照品溶液0.1,0.2,0.4,0.6,0.8,1.0 mL分别置50 mL的量瓶中,用磷酸盐缓冲液(pH6.8)稀释至刻度,摇匀,然后在251 nm处测定吸光度,葛根素浓度与吸光度回归处理,得回归方程:A = 0.0001 + 0.0753C (r = 0.9999),线性范围为1.0~10.0 µg/mL。
3.2.4. 精密度试验
取同一浓度的葛根素对照品溶液,在251 nm处连续测定5次,记录吸光度,计算RSD = 0.97%,说明仪器精密度良好。
3.2.5. 重复性试验
精密称取样品6份,按上述方法制备样品后检查吸光度,计算RSD = 1.05%,表明该方法有良好的
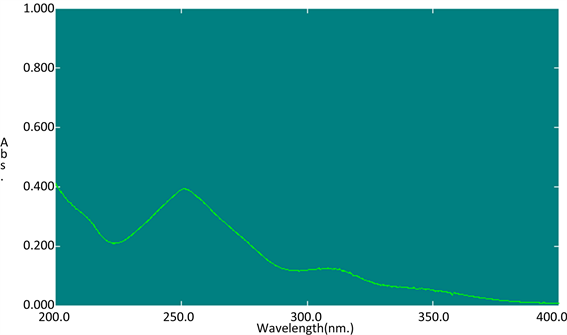
Figure 1. The UV scanning map of puerarin in phosphate buffer (pH6.8)
图1. 葛根素在磷酸盐缓冲液(pH6.8)中的UV扫描图谱
重复性。
3.2.6. 加样回收率试验
分别精密称取葛根素对照品4 mg,5 mg,6 mg各3份,分别加入含葛根素5 mg的样品中(加入量分别相当于样品中葛根素含量的80%,100%,120%),按上述方法制备样品后检查吸光度,计算葛根素的回收率范围为98.60~101.91%,RSD = 1.95%。
3.3. 体外溶出度的测定
照溶出度与释放度测定法(2015版《中国药典》四部通则0931第二法)测定,以磷酸盐缓冲液(pH6.8) 1000 mL为溶出介质,温度37℃ ± 0.5℃,转速为75 r∙min−1,分别于15 min,30 min,45 min取样5 mL (同时补充等量同温介质),滤过,0.8 µm微孔滤膜虑过,加溶出介质稀释成适宜的浓度,作为供试品溶液;另取葛根素对照品适量,用溶出介质分别制成每1 mL含4 µg和8 µg葛根素的溶液,取上述两种溶液,照紫外-可见分光光度法(2015版《中国药典》四部通则0401),在251 nm的波长处分别测定吸光度,计算累积溶出量。
4. 葛根素胶囊处方因素的考察
4.1. 崩解剂种类的考察
选择羧甲基淀粉钠(CNS-Na)、PEG4000、交联聚乙烯吡络烷酮(PVPP) 3种辅料按2.1项下制成胶囊,按2.3项下的方法进行测定45 min内的累积溶出量,结果见图2。
由图2可知,PEG4000辅料作为崩解剂的葛根素胶囊在45 min内累积溶出量明显高于CNS-Na和PVPP。PEG4000虽无崩解性能,但水溶性好,加快了胶囊中葛根素的溶出速度。
4.2. 稀释剂种类的考察
选择糊精、微晶纤维素(MCC)、碳酸钙3种辅料按2.1项下制成胶囊,按2.3项下的方法进行测定45 min内的累积溶出量,结果见图3。
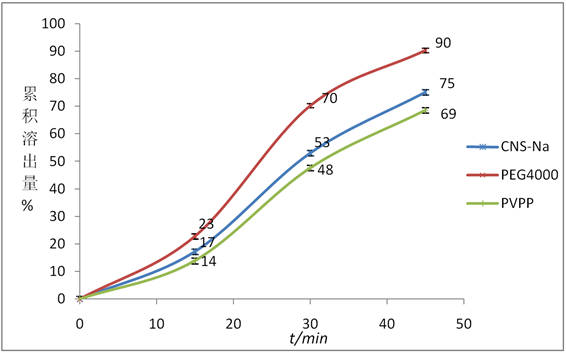
Figure 2. The effect of disintegrating agent on cumulative dissolution of puerarin capsules (n = 3)
图2. 崩解剂种类对葛根素胶囊累积溶出量的影响(n = 3)
由图3可知,3种稀释剂对葛根素胶囊在45 min内累积溶出量的影响不显著。MCC在制剂成型中除了作为稀释剂,其流动性极好,有利于胶囊的填充。
4.3. 辅料比例的考察
MCC和PEG4000按照质量比例为2:1,1:1,1:2按2.1项下制成胶囊,按2.3项下的方法进行测定45 min内的累积溶出量,结果见图4。
由图4可知,MCC和PEG4000辅料比例为1:1和1:2的葛根素胶囊在45 min内的累积溶出量均比MCC和PEG4000比例为2:1的高;辅料比例为1:1和1:2在45 min的累积溶出量无显著差异,但在45 min之前,辅料比例为1:1均比1:2的累积溶出量高,有利于药物在体内及时吸收。

Figure 3. The effect of thinners on the cumulative dissolution of puerarin capsules (n = 3)
图3. 稀释剂种类对葛根素胶囊累积溶出量的影响(n = 3)

Figure 4. The effect of different excipients on cumulative dissolution of puerarin capsules (n = 3)
图4. 不同辅料比例对葛根素胶囊累积溶出量的影响(n = 3)
根据处方筛选结果,胶囊处方组成比例为:葛根素磷脂复合物为60%,MCC比例为19.5%,PEG4000比例为19.5%,微粉硅胶为1%。
5. 结论
建立体外溶出度标准的目的是保证药品批间质量的一致性,并提示可能的体内生物利用度问题。普通口服固体制剂可采用下列两种溶出度控制方法:(1) 单点检测:可作为常规的质量控制方法,适用于快速溶出的高溶解性药物制剂。(2) 两点或多点检测:可反映制剂的溶出特征;作为某些类型药物制剂的常规质量控制检验 [8] 。葛根素为溶解性较差的药物,应采用两点或多点检测,本实验采用三点检测来建立葛根素胶囊体外溶出度标准,建立的体外紫外分析方法方便、科学、可行。
实验通过对葛根素胶囊体外溶出度影响因素进行考察,确定了处方组成为葛根素磷脂复合物的比例为60%,MCC比例为19.5%,PEG4000比例为19.5%,微粉硅胶为1%。15 min、30 min、45 min的累积溶出量分别为27%、78%、98%,具有良好的体外溶出特性。
葛根素胶囊具有制备工艺简单,体外溶出速度快的特点,口服用药后可迅速释放进入消化液,吸收进入体内,能及时起到预防治疗心血管疾病的目的。
基金项目
2016年南宁市科技计划项目(20163160)。